PeptiGel Resources
PeptiGel® Product Support Literature
Application Notes
Protocols
Frequently asked questions (FAQs)
Highlighted Publications
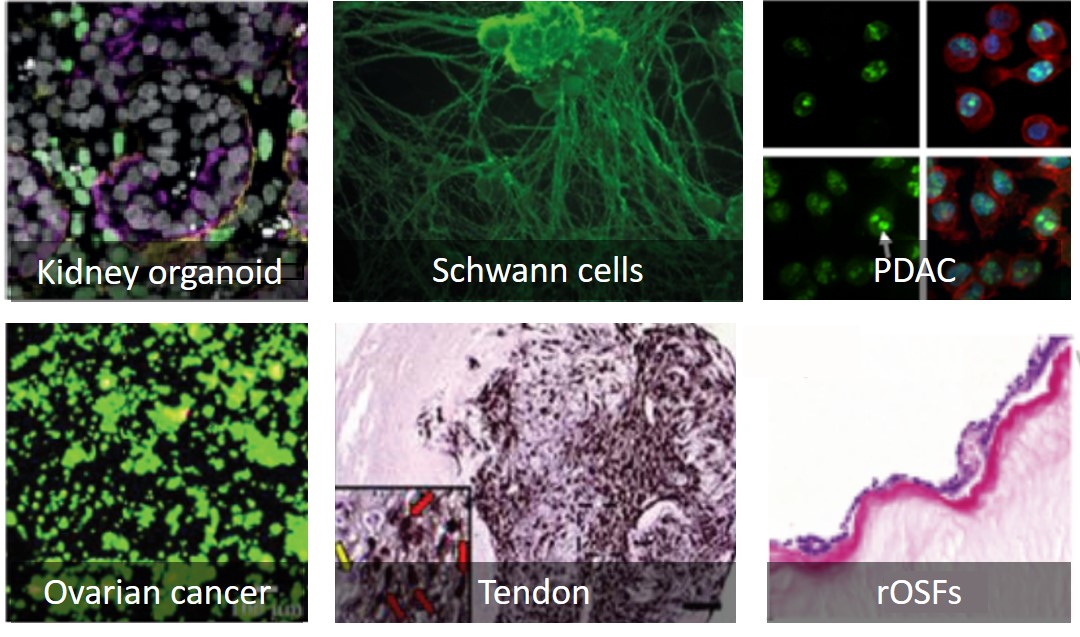
Many types of cells and tissues have been cultured with PeptiGel®
Examples of cells grown with PeptiGel® hydrogels. PDAC= pancreatic ductal adenocarcinoma, rOSFs = rat oesophageal stromal fibroblasts. For further details, please see the PeptiGel resources page.
PeptiGel characteristics, demonstrated cell compatibility and publications
Click on the cell type in the table for a link to the publication.
Gel type characteristics and demonstrated compatible cells |
||||
|
PeptiGel® Type |
Charge |
Stiffness (kPa) |
Biomimetic Functionality |
Demonstrated Compatible Primary Cells and Cell Lines |
|
Alpha 1 |
Neutral | 3 - 5 | None |
Fibroblast 3T3, Kidney HEK293, Schwann cells, |
| Alpha 2 | +1 | 6 - 8 | None |
bone marrow model (haemopoietic stem cells (HSCs) and mesenchymal stem cells (MSCs)), iPSC-derived cardiomyocytes (in vivo administration), Prostate: PC3 pNT2, |
|
Alpha 2 Plus |
+1 | 6 - 8 | RGD & GFOGER | Neuronal (in vivo administration) |
| Alpha 4 | +2 | 0.7 - 1.3 | None |
Bone marrow-derived stem cells, Fibroblasts: 3T3 + L929, HUVECS Muscle C2C12, |
| Alpha 4 Plus | +2 | 0.7 - 1.3 | RGD & GFOGER |
iPSC-derived cardiomyocytes (in-vivo administration), Stromal fibroblasts, |
| Alpha 8 | -3 | ~ 0.25 | None | Hepatic Cells |
| Delta 1 | Neutral | 3 - 5 | None | Bone marrow model (haemopoietic stem cells (HSCs) and mesenchymal stem cells (MSCs)), |
| Gamma 2 | +1 | ~ 1.5 | None | |
| Gamma 2 Plus | +1 | ~ 2.5 | RGD & GFOGER | Neuronal |
| Gamma 4 | +2 | 0.175 - 0.35 | None | For cells requiring a softer environment, including primary cells, immortalized cells and stem cells |
| Gamma 4 Plus | +2 | 0.175 - 0.35 | RGD & GFOGER | Spheroids and organoids |
|
|
||||
The stiffness values given here are indicative and have been measured by shear rheometry after 24 incubation in DMEM media.
Choosing a PeptiGel® for your cells |
||||
Primary Cells |
||||
|
Origin |
Cell Type |
Species |
Catalog products |
Custom products |
|
Blood |
Monocytes | Murine | Alpha 1 |
|
| Bone marrow | Haemopoetic stem cells | Human | Alpha 2, Delta 1 | |
| Bone | Osteoblasts | Human | Alpha 1 | |
|
Breast |
Mammary epithelial cells | Human | Alpha 4 | |
| Breast | Breast Cancer explant | Human | Alhpa 1 | |
| Cartilage | Chondrocytes | Human | Alpha 1 | |
| Cartilage | Chondrocytes | Bovine | Alpha1, Alpha 2 | Alpha 3 |
| Dermal | Fibroblasts | Human | Alpha 1, Alpha 4 | Alpha 4 YIGSR |
| Oesophagus | Stromal fibroblasts | Rat | Alpha 4, Alpha 4 PLUS | Alpha 5 |
| Intervertebral disc | Nucleus pulposus cell | Bovine | Alpha 1 | |
| Kidney | Fibroblast | Human | Gamma 2, Alpha 2 PLUS | |
| Muscle | Smooth Muscle | Mouse | Alpha 1 PLUS | |
| CNS | Neuronal cell | Rat | Alpha 3 | |
| CNS | Neonatal Schwann | Human | Alpha 1 | |
| Oesophagus | Oesophageal epithelial | Mouse | Alpha 1 | |
| Skin | Keratinocytes | Human | Alpha 1 | |
| Spinal cord | Dorsal root ganglion neurons | Rat | Alpha 2, Alpha 4, Gamma 2 | |
| Synovial Membrane | Synovial cells | Rat | Alpha 4 | |
Cell lines |
||||
| Breast | MCF7 | Human | Alpha 4 | Alpha 4-Lam 111 |
| Breast | MCF10a | Human | Alpha 4 | |
| Breast | EPH4 | Mouse | Alpha 1, Alpha 4 | |
| Breast | MDAMB231 | Human | Alpha 4 | |
| Embryo | 3T3 fibroblast | Mouse | Alpha 1, Alpha 2, Alpha 4 | |
| Fibroblast | L929 | Human | Alpha 2 | Alpha 5 |
| Kidney | HEK293 | Human | Alpha 1, Alpha 2, Alpha 4 | Alpha 5 |
| Liver | HepG2 | Human | Alpha 2 | Delta 6 |
| Muscle | C2C12 | Human | Alpha 4 | |
| Ovary | A2780 | Human | Alpha 4, Alpha 4 PLUS | |
| Pancreas | Suit-2 | Human | Alpha 2, Gamma 2 | |
| Prostate | PC3 | Human | Alpha 2 | |
| Prostate | LNCaP | Human | Alpha 2 | |
| Prostate | PNT2 | Human | Alpha 2 | |
| Umbilical vein | HUVEC | Human | Alpha 4 | |
Stem cells |
||||
| Adipose | Adipose-derived stem cells | Human | Alpha 1, Alpha 2 | |
| Bone marrow | Bone marrow-derived haemopietic stem cells | Human | Alpha 1, Delta 1, Alpha 4 | |
| Bone marrow | Mesenchymal stem cells | Human | Alpha2, Delta 1 | |
| Intestine | ATZ stem cells | Human | Alpha 4-IKVAV, Alpha 4-YIGSR | |
| iPSCs | Cardiomyocytes | Mouse | Alpha 2, Alpha 4 | Alpha 2-Lam111 |
| Somatic cell | iPSCs | Human | Alpha 1, Alpha 4 | Alpha 5 |
Organoids and co-culture |
||||
| iPSCs | Kidney | Human | Alpha 4 | |
| Crypts | Colon | Mouse | Alpha 4, Gamma 2, Alpha 4 PLUS | Alpha 5 |
| Liver | Mixed | Pig | Alpha 2 | |
| ASC + dorsal root ganglion | Alpha 2 | |||
| Epithelial cells + fibroblasts | Alpha 4, Alpha 5 | |||
In vivo |
||||
| Neuronal | Model of intracerebellar haemorrhage | Rat | Alpha 2 plus | |
| Cardiac | Cardiac delivery of iPSC-derived cardiomyocytes | Mouse | Alpha 4 plus | |
| Cardiac | Rat Cardiac Progenitor cells | Rat | Alpha 1 plus | |
Webinar: How to Successfully Make the Switch to Synthetic Peptide Hydrogels
Professor Aline Miller (University of Manchester) and Sebastian Doherty-Boyd (University of Glasgow) discuss the difficulties in producing consistent cell culture results using traditional biomaterials, particularly animal-derived matrices with their inherent variability. They also discuss how to gain greater control over 2D and 3D cell culture with PeptiGels and cover specific examples of using PeptiGels to generate 3D tissue and disease models. Published: September 2023
For technical support, please get in touch with us by email at [email protected].
Appendix: Application Note Abstracts
1. The use of PeptiGels® in the development of gastro-intestinal organoids
Organoid formation in preclinical studies requires the use of an extracellular matrix (ECM), such as Matrigel™. However, these ECMs are often derived from a tumorigenic source which limits the translational data and scalability adoption of organoids. Offering a synthetic and animal-free solution, Dr. Dammy Olayanju (Northwick Park Institute for Medical Research) applied PeptiGels® to provide a disease-free ECM which successfully supported the growth of fully formed liver/gastro-intestinal organoids for up to 27 days. The successful growth of these organoids allowed for reproducible and scalable results to be achieved, opening the potential for PeptiGels® to provide more reliable preclinical studies for pharmacological and toxicology studies.
2. Self-assembling peptide hydrogel recapitulates in-vitro mechanisms of breast cancer progression
The tumour microenvironment (TME) is a complex network of cancer cells, other cell types, and extracellular matrix (ECM) molecules, and is essential for the development of tumours. To study cancer initiation and progression, and for the development of drugs targeting the TME in vitro, these components must be replicated. PeptiGel® stiffness can be modulated to mimic breast cancer tissue, and they can also be functionalised with ECM proteins and cell recognition sequences to increase cell proliferation and survival. Olga Tsigkou’s research group (University of Manchester) have used PeptiGels® to support the growth of a 3D tumour model using MCF-7 breast cancer cells, HUVEC, and MSC cells. This is a very promising solution for studying 3D in vitro tumour models using animal-free materials, for drug discovery applications.
3. Delivering stem cell therapies for nerve repair
Peripheral nerve injuries (PNI) are a common consequence of trauma to the upper and lower limbs. Current treatment involves autografting nerves which can cause donor site morbidity and loss of sensitivity. Schwann cells are key cells involved in nerve regeneration, however obtaining them requires harvesting healthy patient nerves. As an alternative approach to the regeneration of PNI, Dr. Adam Reid’s group (University of Manchester) successfully used PeptiGels® as synthetic scaffolds for the culture and chemical differentiation of human derived-adipose stem cells toward a Schwann cell-like phenotype. This research has proven the capabilities of PeptiGels® in delivering cells for an alternative approach to the regeneration of peripheral nerves, and in future applications for bioengineered nerve grafts.
4. PeptiGels® can mimic both healthy and pancreatic cancer tumour tissue properties
The extracellular matrix of different organs and cellular settings have very different chemical properties. In pancreatic ductal adenocarcinoma (PDAC), acidic fibrous tissue is present around the tumour which hampers drug delivery and impacts cancer cell survival and proliferation. PeptiGel® pH and stiffness can be modulated (1-20 kPa) to mimic the acidic fibrous cancer tissue, as well as healthy tissue, making them very suitable for applications in mechanobiology and cancer. Dr Armando Del Rio Hernandez’s research group (Imperial College London) have used PeptiGels® to mimic the mechanical and chemical environment of both healthy and cancer tissue. Results showed the tumour mimicking PeptiGel® (stiff and acidic) to induce a biomechanical response in PDAC suit-2 cells, resulting in an increased proliferation (expression of Ki67).
5. Successful growth of kidney organoids using PeptiGels®
Induced pluripotent stem cell derived kidney organoids offer valuable insights into how the organ develops in the body. However, a gap remains in the understanding surrounding the optimal conditions required for the generation of kidney organoids that fully mimics the in vivo organ. PeptiGel® and its tuneable properties offer an ideal environment in which the organoid is developing to be optimised. This has allowed Niall Treacy (UCD Conway Institute of Biomolecular and Biomedical Research) to successfully grow kidney organoids in an animal-free synthetic environment. By bridging the gap in the understanding of the optimal conditions for organoid growth, PeptiGels® offer an increased understanding of kidney organoid in-vitro modelling for toxicity testing and drug discovery.
6. Development of PeptiGel® based 3D in-vitro model of epithelial ovarian cancer
Epithelial Ovarian Cancer (EOC) is a leading cause of female cancer deaths worldwide. Understanding its mechanisms and metastasis requires the development of effective experimental models of EOC and its interaction with the surrounding microenvironment. Here, the effects of biomaterial stiffness and ECM protein functionalization on ovarian cancer cell growth were studied. Promising results were observed in PeptiGel® Alpha 4, Alpha 4-RGD, and Alpha 4-GFOGER hydrogels. PeptiGel® Alpha 4 showed optimal long-term 3D growth, while Alpha 4-GFOGER promoted earlier cellular growth. All hydrogels supported ovarian cancer cell growth at day 28. This research enhances the understanding of EOC pathology and microenvironment interactions for potential therapeutic advancements.
7. Engineering a cell-hydrogel-fibre composite to mimic the structure and function of the tendon synovial sheath
Poor recovery from flexor tendon injuries often results in pain, disability, and reduced hand function. Traditional tendon repairs using sutures can lead to poor function due to adhesions between the tendon and surrounding tissues. This study explores a novel approach using PeptiGel® Alpha 4 seeded with synoviocytes and combined with an electrospun nanofibrous poly (ε-caprolactone) mesh to create a bilayer biomembrane. The biomembrane demonstrated potential in reducing post-operative adhesions, restoring tendon gliding ability, and supporting synovial sheath repair. Biocompatibility tests showed favourable results with synoviocytes, suggesting improved long-term tendon functionality with this approach.
8. PeptiGels® can prevent oesophageal strictures and promote the regeneration of healthy tissue
Barrett’s oesophagus, if left untreated, often leads to oesophageal cancer, the 8th most common cancer in the world. Current treatments to remove the pre-cancerous cells can damage healthy cells and lead to fibrotic strictures. Prof Julie Gough’s research group (University of Manchester) have used PeptiGels® (peptide hydrogels) to co-culture rat oesophageal stromal fibroblasts, and mouse oesophageal epithelial cells to support the formation of a functional epithelial sheet. PeptiGels® are mucoadhesive and can be sprayed endoscopically directly to the treatment areas, making them a promising solution towards treatment for stricture management in Barrett’s oesophagus, and a perfect solution for tissue regenerative scaffolds in general.
9. Using PeptiInks® to print mammary epithelial cell laden constructs with high structural integrity
3D bioprinting is an emerging field in drug discovery, cancer, and regenerative medicine. The key limitation however is the development of an optimal bioink that is able to print complex structures with geometrical accuracy, tuneable mechanical stiffness, and long-term cell viability. Dr. Marco Domingo’s group (University of Manchester) used PeptiGels® with varying stiffness to print mammary epithelial cell laden constructs with high structural integrity using an extrusion based bioprinter. The printed samples were subsequently cultured for 7 days. PeptiGels® have shear thinning properties, hence they can be injected, printed, and recover their structural integrity immediately after the shear is removed. PeptiGels® also have tuneable mechanical properties and they can promote cell attachment, proliferation, and differentiation, making them suitable bioinks for 3D bioprinting applications.