Cell Guidance Systems provides professional karyotype analysis for research cells and cell lines. We typically analyse a minimum of 20 G-banded metaphase spreads per sample, delivering a detailed report with publication-quality images. Standard turnaround is 7 -10 business days, with an express option available for urgent projects. We routinely process human and mouse samples and are experienced in karyotyping for a range of other species.
Why Karyotype Your Cell Lines?
Chromosomal abnormalities accumulate during prolonged cell culture, particularly in iPSC and embryonic stem cell lines. These karyotypic changes can alter gene expression, affect differentiation potential, and compromise experimental reproducibility. For iPSC-based research, routine karyotyping is considered essential quality control; the International Society for Stem Cell Research (ISSCR) and regulatory bodies including the FDA and EMA require evidence of genomic stability for cell lines intended for therapeutic applications.
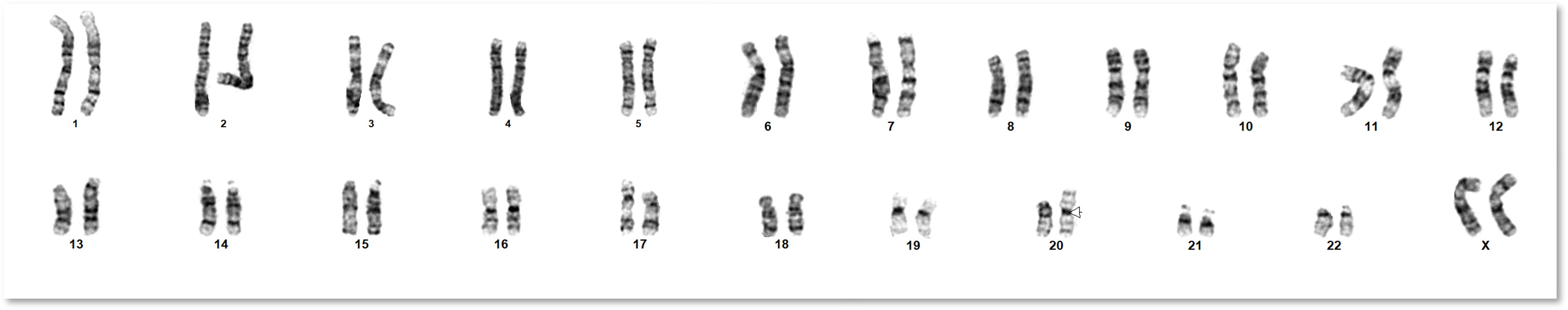
G-banding karyotype analysis remains the standard method for detecting numerical and large structural chromosomal abnormalities. It identifies gains, losses, translocations, inversions, and other rearrangements across all chromosomes in a single assay. Common findings in iPSC and ESC cultures include abnormalities of chromosomes 1, 12, 17, 20, and X. Karyotype analysis can also detect de novo abnormalities that are often missed by targeted methods such as qPCR panels.
We recommend karyotyping:
- After reprogramming, to confirm the iPSC line has a normal karyotype before banking
- Every 10–15 passages during routine culture
- Before functional experiments, differentiation, or downstream applications
- As part of cell line authentication for publication or regulatory submission
- When unexpected changes in growth rate or morphology are observed
What We Deliver
Every analysis includes a comprehensive report with the karyotype designation in ISCN nomenclature, a description of any abnormalities, and at least one publication-quality image of a representative metaphase spread. For unstable or highly abnormal samples, we characterise sufficient cells to identify all clonal abnormalities. Example reports are available on each service page.






Karyotype analysis for human cells Live (BSL1) or fixed (BSL1/BSL2) human cell lines, including iPSCs, ESCs, and primary cultures. Standard and express options are available.
Karyotype analysis for mouse cells Live (BSL1) or fixed (BSL1/BSL2) mouse cell lines.
Karyotype analysis for porcine cells Live (BSL1) or fixed (BSL1/BSL2) pig cell lines.
Karyotype analysis for bovine cells Live (BSL1) or fixed (BSL1/BSL2) cow cell lines.
Karyotype analysis for cell lines of other species Live (BSL1) or fixed (BSL1/BSL2). Other species include porcine, bovine, ovine, canine. Please enquire for any other species.
Array Genomic Hybridisation (AGH) High-resolution chromosomal analysis using Infinium Global Screening Array v3.0. Ideal for identifying material of unknown origin. Human samples only.
Common species:
We also have capabilities for
- Cow karyotyping (bovine karyotyping)
- Pig karyotyping (porcine karyotyping)
- Dog karyotyping (canine karyotyping)
- Sheep karyotyping (ovine karyotyping)
- Goat karyotyping (caprine karyotyping)
- Chicken karyotyping (gallus karyotyping)
- Horse karyotyping (equine karyotyping)
Contact us for further details or if your species isn't listed.
Service levels
We accept 2 types of samples in our laboratories:
- Live cells (BSL1 only): cells arrive in culture in a T25 flask (available for UK and Europe only)
- Fixed cells: cells arrive fixed in a 2 mL screwcap tube - we will provide detailed instructions on how to prepare the cells
Please do not send any samples without booking in advance
Number of cells analyzed:
- Normal cells: 20 cells are analyzed per sample as a minimum. This number can be increased to your requirements
- For unstable cancer/highly abnormal samples: sufficient cells will be characterized to identify clonal abnormalities
Reporting times:
- Standard service: 7-10 business days (human or mouse)*
- An Express service is available: please contact us to check availability and turnaround before ordering
*Please note that reporting times may be longer during peak periods. This guidance is for human and mouse. Other species may take longer. You will be advised of any expected delays when placing an order.
For more details, and a sample report see Services Description
Karyotyping vs Array Analysis
G-banding karyotype analysis and array genomic hybridisation provide complementary information. Karyotyping detects balanced rearrangements (translocations, inversions) that arrays cannot see. Array analysis offers higher resolution for smaller copy number changes. For routine iPSC and cell line QC, G-banding is typically the appropriate first-line test. We recommend array analysis when karyotyping identifies material of unknown origin, or when higher-resolution detection is needed.
Frequently Asked Questions
How many cells do you analyse per sample?
We analyse a minimum of 20 G-banded metaphase spreads. For unstable or highly abnormal samples, we characterise sufficient cells to identify all clonal abnormalities.
How often should I karyotype my iPSC lines?
We recommend karyotyping every 10–15 passages during routine culture, plus after reprogramming, before banking, and before experiments or therapeutic applications.
Do you offer an express service?
Yes. Contact us to check availability and turnaround before ordering.
What species can you karyotype?
We routinely process human and mouse samples. We also offer karyotyping for bovine, porcine, ovine, caprine, gallus, canine, and equine cells. Contact us if your species is not listed.
Can you process BSL2 samples?
We accept BSL2 human samples as fixed cells (no screening required).
Please note that we do not offer clinical services for patients.
If you have any questions, please contact us by email at [email protected]. We will respond within one business day.