Primary human hepatocytes now available

Cell Guidance Systems is pleased to announce the availability of primary human hepatocytes for use in in-vitro toxicology assays.
The development of drugs requires a range of assays to evaluate safety. Many of the fundamental safety tests which small molecule drugs undergo are focussed on the liver as it is the primary site of drug metabolism. Although significant progress has been made with iPSC-derived hepatocytes, they can't accurately replicate primary human hepatocytes which remain the gold standard.
Small molecules are metabolized by enzymes in the liver in a process known as biotransformation which has two key phases. The first phase, which is largely driven by endoplasmic reticulum-bound cytochrome P450 enzymes, yields polar, water-soluble metabolites. These metabolites are often still active. In the second phase, metabolites have endogenous hydrophilic groups added to form water-soluble compounds that can be secreted from the body. A further Phase III can produce additional metabolism and secretion of the target substance to enhance excretion.
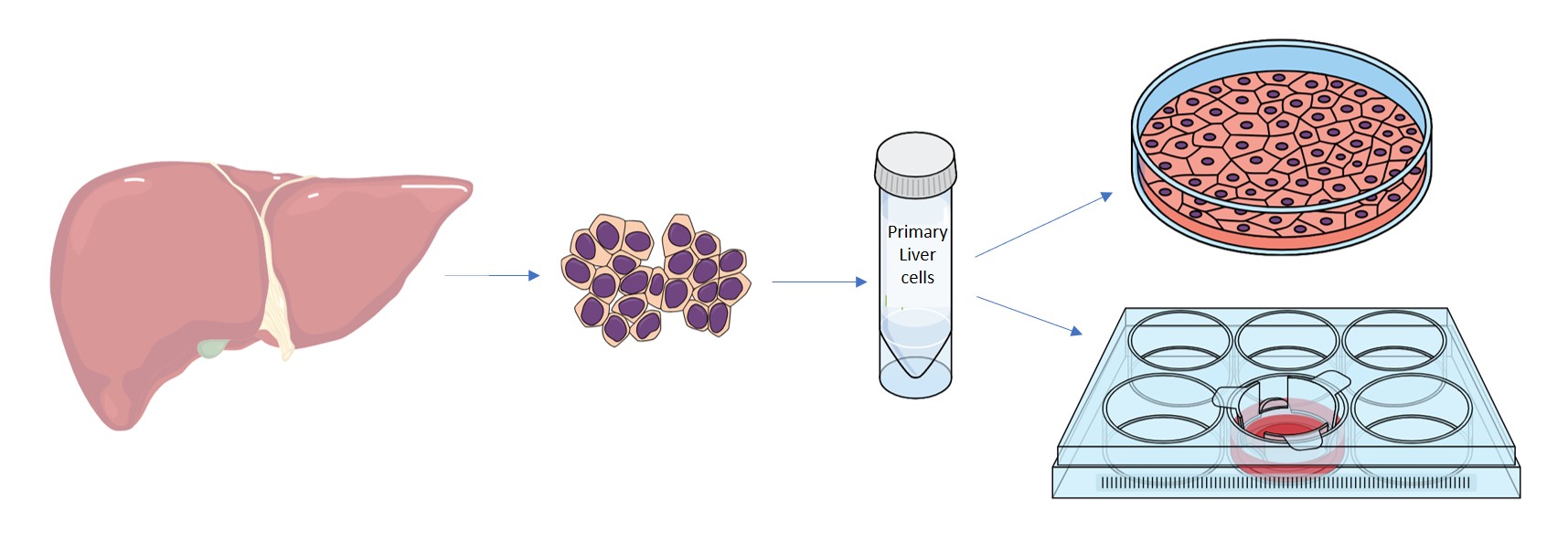
The primary human hepatocytes offered by Cell Guidance Systems are ethically sourced from patients undergoing surgery or from transplant organs that could not be used. The hepatocytes are gently removed from the tissues and rapidly frozen in liquid nitrogen where they are constantly maintained during storage and transport, right up to the point of receipt by the customer.
Cell Guidance Systems offers three grades of liver cells suitable for a range of different applications.
- Plateable – ability to form a monolayer
- Suspension – grow in suspension
- Spheroid – form spheroids in culture
Both the Plateable and Suspension-grade hepatocytes are comprehensively characterized for metabolic activity. The Spheroid grade cells are quality controlled for their ability to form spheroids in culture.
Further information, including sample Certificates of Analysis, can be found on the product page.
IMAGE: Hepatocytes for toxicology assays. Cell Guidance Systems.